SUSTOL—the only 5-HT3 RA with polymer technology that is engineered to last for ≥5 days*1-7
- Granisetron is a proven, effective treatment option for the prevention of CINV, and its therapeutic benefits are well established1,2
- The unique polymer technology of SUSTOL is engineered to allow for a sustained and controlled release of granisetron to prevent CINV for ≥5 days1

GRANISETRON/POLYMER COMBINATION
- SUSTOL is engineered using a unique polymer technology that enables a controlled release of the otherwise shorter-acting granisetron1,2
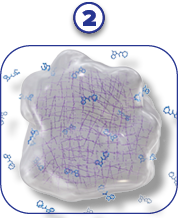
RELEASE OF
GRANISETRON
- After subcutaneous injection, the polymer undergoes hydrolysis and delivers granisetron in a controlled, sustained release for ≥5 days1,2

POLYMER
HYDROLYZES
- After the granisetron has been released, the polymer hydrolyzes and is eliminated from the body1,2
- SUSTOL is indicated for the prevention of CINV due to MEC and AC combination chemotherapy.1
5-HT3 RA=5-hydroxytryptamine 3 receptor antagonist; AC=anthracycline/cyclophosphamide; CINV=chemotherapy-induced nausea and vomiting; MEC=moderately emetogenic chemotherapy; PK=pharmacokinetics.